Sleep
-
$0.00
VOQUILY® (melatonin – immediate release) 2mg
VOQUILY® is indicated for:
Sleep disorders in children and adolescents aged 6 to 18 years with neurodevelopmental disorders, including autism spectrum disorder (ASD) and attention deficit hyperactivity disorder (ADHD), where sleep hygiene measures have...$0.00
VOQUILY® (melatonin – immediate release) 3mg
VOQUILY® is indicated for:
Sleep disorders in children and adolescents aged 6 to 18 years with neurodevelopmental disorders, including autism spectrum disorder (ASD) and attention deficit hyperactivity disorder (ADHD), where sleep hygiene measures have...$0.00
VOQUILY® (melatonin – immediate release) 5mg
VOQUILY® is indicated for:
Sleep disorders in children and adolescents aged 6 to 18 years with neurodevelopmental disorders, including autism spectrum disorder (ASD) and attention deficit hyperactivity disorder (ADHD), where sleep hygiene measures have...Pain Relief
-
$0.00
RELPAX
Relpax® is indicated for:
Acute treatment of migraine headache with or without aura.Relpax® 40mg (eletriptan hydrobromide)
...$0.00
ZEA RELIEF KUNZEA PAIN RELIEF CREAM
Kunzea Pain Relief Cream (AUST L 360 331) is an anti-inflammatory cream that contains naturally derived active ingredients, specifically formulated for muscle and mild joint pain.
...Gastro Health & Digestion
-
$0.00
GLUTEGUARD – GLUTEN DEGRADING ENZYME
GluteGuard tablets provide clinically supported protection for those with coeliac disease and other medically diagnosed gluten sensitivities from symptoms of unintentional gluten ingestion.
...Cold & Flu
-
$0.00
MAXIGESIC® COLD & FLU HOT DRINK
Maxigesic® Cold & Flu Hot Drink combines the power of paracetamol and ibuprofen in a unique formulation to provide fast, effective double-action cold & flu relief.
...Medication lubricant
-
$0.00
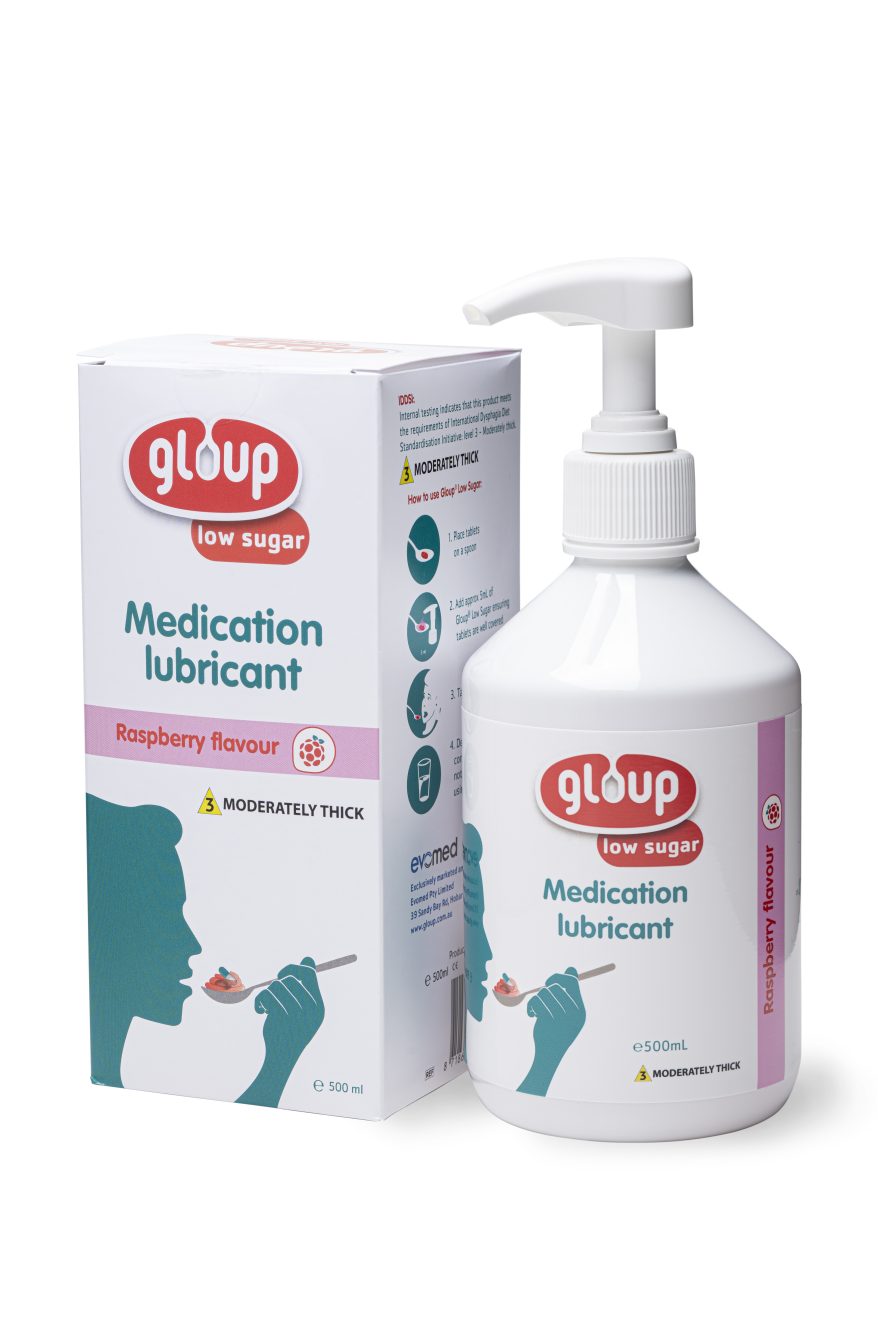
GLOUP® (Pump Bottle)
Gloup Medication Lubricant makes tablets easier to swallow, reduces the need to crush, and is recommended by multiple clinical guidelines*.
...$0.00
GLOUP® (Tube)
Gloup Medication Lubricant makes tablets easier to swallow, reduces the need to crush, and is recommended by multiple clinical guidelines*.
...Asthma
-
$0.00
EASYHALER® DRY POWDER INHALER
Easyhaler® is a carbon neutral dry powder (DPI) respiratory inhaler available on the PBS for the treatment of COPD and asthma¹. Salflumix (salmeterol/fluticasone propionate) Easyhaler® is available in 500µ/50µ and 250µ/50µ². Bufomix Easyhaler® (budesonide/formoterol fumarate...
Pregnancy
-
$0.00
Out of stockNATALIS PREGNANCY MULTIVITAMIN
Natalis is an Australian Made pregnancy multivitamin formulated to support the increased nutritional needs from pre-conception to pregnancy and breastfeeding as well as support baby’s healthy development.
Containing the same amounts of key...
Dietary supplements
-
$0.00
Out of stockOSTELIN VITAMIN D 7000IU
Ostelin Specialist Range Vitamin D 7000IU is a convenient one-a-week formula made in Australia from carefully selected high quality ingredients.
...$0.00
Out of stockVITAMIN D LIPO-SACHETS
Vitamin D is crucial for immune system function and overall health, including bone health. With Lipo-Sachets® liposomal vitamin D3, your patients can enjoy the benefits of this essential vitamin with up to 4x higher...
Dermatology
-
$0.00
Out of stockAKLIEF
Aklief is indicated for the topical treatment of Acne Vulgaris of the face and/or the trunk in patients from 12 years of age and older, when many comedones, papules and/or pustules are present.
...$0.00
ANTHELIOS INVISIBLE FLUID SUNSCREEN SPF 50+
A lightweight daily facial sunscreen made for sensitive skin providing very high UVA/UVB protection while delivering an invisible finish.
This fragrance-free, facial sunscreen is suitable normal to combination skin, specifically designed...
$0.00
CERAVE MILD ECZEMA-PRONE SKIN KIT
Developed with dermatologists, all CeraVe skincare products are formulated with 3 essential ceramides, which are an essential part of a healthy skin barrier. The CeraVe range is designed with moisturising MultiVesicular Technology [MVE]...
$0.00
EFFACLAR ANTI-ACNE KIT
Anti-Acne Kit to target blemishes and imperfections for oily, acne prone skin.
EFFACLAR HYDRATING KIT INCLUDES:
EFFACLAR DUO+M CLEANSER 15ML
EFFACLAR DUO +M 3ML
ANTHELIOS INVISIBLE FLUID...
$0.00
Out of stockQV DERMCARE
QV Dermcare is a specialised range of skincare for eczema prone skin, with products developed to support the skin barrier, help improve hydration and relieve dryness and discomfort.
Clinically tested to help manage mild to...
$0.00
TISSUE REPAIR
A new standard in aftercare treatment. Clinically validated to deliver up to 40% improvement in skin quality. Accelerates healing.
...Gout
-
$0.00
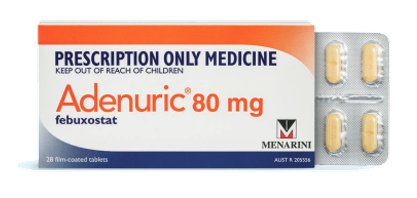
ADENURIC® (febuxostat)
ADENURIC® (febuxostat) is indicated in the treatment of chronic symptomatic hyperuricaemia in conditions with urate deposition in adults with gout.
Prescription Medicine, Doctor Requests Only.Febuxostat 80 mg (4 tablets)NOTE: Only sample (4 tablets)...Mens health
-
$0.00
Out of stockTESTOGEL®
Testogel® is indicated for use as testosterone replacement therapy for male hypogonadism when testosterone deficiency has been confirmed by clinical features and biochemical tests. To request a copy of the Product Information or to report adverse...
Breakthrough Cancer Pain
-
$0.00
ABSTRAL (Fentanyl citrate)
Samples of this product cannot be sent out, however the Manufacturer has offered the options below for you to select from. This product does not count towards your minimum cart size
ABSTRAL Fentanyl...
Eye Care
-
$0.00
NOVATEARS®
NovaTears® is a unique preservative-free eye lubricant and tear film stabiliser in a multi-dose bottle specifically designed for the relief of Evaporative Dry Eye Disease.
...Infant Formula
-
$0.00
Out of stockEASY-DIGEST GOAT MILK FORMULA STAGE ONE
Exclusively formulated for babies aged 0 to 6 months, or when transitioning from breastfeeding.
Bubs Easy-Digest Goat Milk Infant Formula is derived from naturally occurring A2 Goat Protein and contains natural goat milk of...$0.00
Out of stockEASY-DIGEST GOAT MILK FORMULA STAGE TWO
Exclusively formulated for babies aged 6 to 12 months, or when transitioning from breastfeeding.
Bubs Easy-Digest Goat Milk Follow-on formula is derived from naturally occurring A2 Goat Protein and contains natural goat milk of...Iron Deficiency
-
$0.00
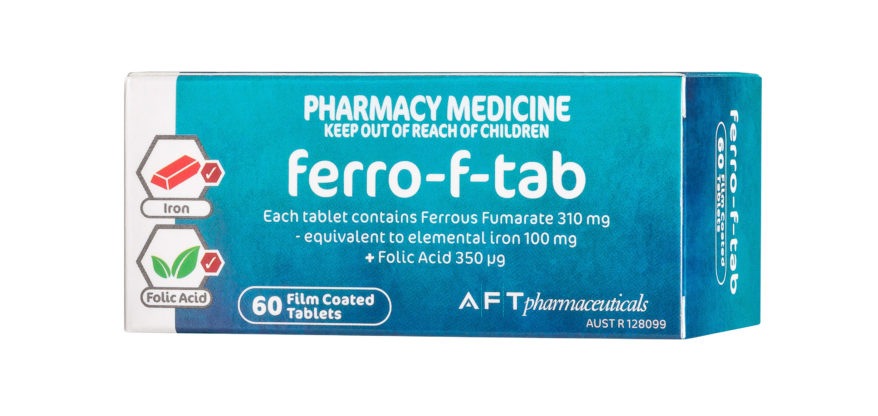
FERRO F TAB
The flexible Ferro-f-tab dosage enables you to effectively treat patient iron and folic acid deficiency and helps manage the common digestive side effects that may result from treatment.
Recommend Ferro-f-tab for iron and folic...$0.00
FERRO TAB
The flexible Ferro-Tab dosage enables you to effectively treat patient iron deficiency and helps manage the common digestive side effects that may result from treatment.
Dosage (adults):
Prevention ο
...Womens health
-
$0.00
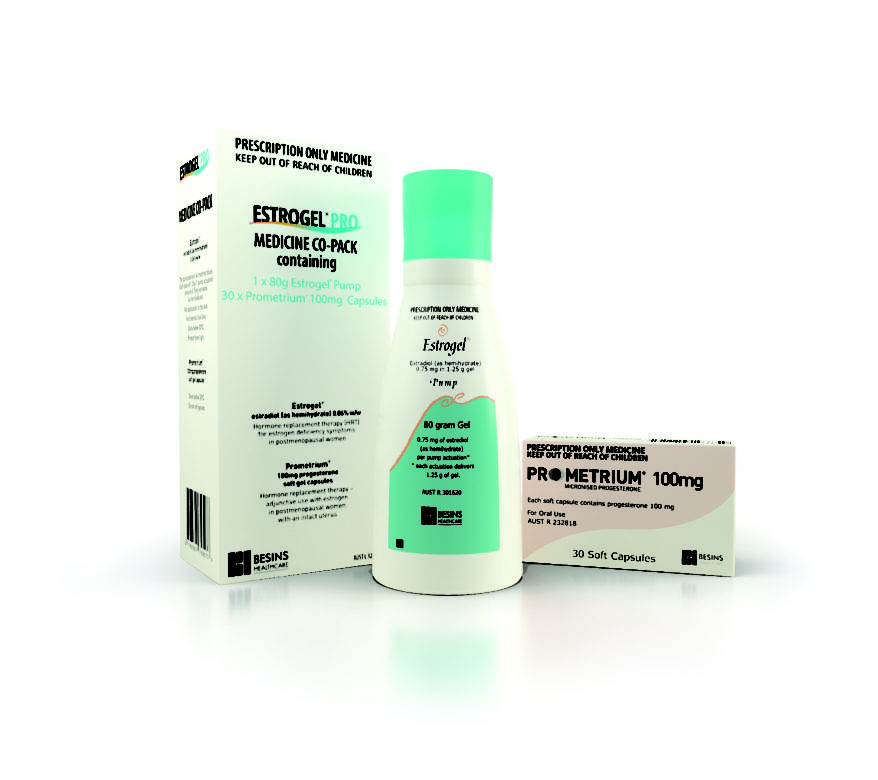
ESTROGEL® PRO
Samples of this product cannot be sent out, however the Manufacturer has offered the options below for you to select from. This product does not count towards your minimum cart size.Estrogel Pro Medicine...
$0.00
Out of stockFEMME-TAB (30/150)
Femme-Tab (ED 30/150) oral contraceptive available on the PBS.
$0.00
PROMETRIUM®
Samples of this product cannot be sent out, however the Manufacturer has offered the options below for you to select from. This product does not count towards your minimum cart size.
Prometrium (micronised progesterone)...
$0.00
SANDRENA®
SANDRENA® Transdermal Estradiol Gel
Samples of this product are not available. Orion Pharma offers a Sandrena Information Pack which contains:
- 1 Sandrena Demonstration pack of 28 sachets with no active...
$0.00
SLINDA®
Slinda® (drospirenone 4 mg): The only progestogen-only contraceptive pill with a 24-hour missed pill window1
...$0.00
UTROGESTAN® 200mg
Utrogestan (micronised progesterone) 200mg x 42 soft vaginal capsules.
Pack size foil of 7 capsules.
...$0.00
UTROGESTAN® 300mg & 400mg
Samples of this product cannot be sent out, however the Manufacturer has offered the options below for you to select from. This product does not count towards your minimum cart size.
Utrogestan (micronised progesterone) 300mg/400mg x...
Probiotics
-
$0.00
BIOGAIA GASTRUS PURE ACTION 2-PACK
Helps relieve symptoms of medically diagnosed IBS.
...
Reduce abdominal discomfort, wind and bloating.
Helps support normal bowel function.
Assists with maintenance of good gut flora during antibiotic use.$0.00
Out of stockBIOGAIA PRODENTIS FOR ADULTS AND KIDS
BioGaia Prodentis lozenges is a daily oral probiotic to support healthy gums and teeth. Each lozenge features two patented bacterial strains that support a balanced oral microbiota. When combined with regular brushing and flossing, BioGaia...
$0.00
BIOGAIA PROTECTIS BABY PROBIOTIC DROPS 2ML
Out of stockBioGaia Protectis Baby Drops are a natural and effective baby probiotic formulated to relieve symptoms of infant colic.
Each request includes
- 3 x BioGaia Protectis Drops 2ml sample size
- 3 x...
$0.00
BIOGAIA PROTECTIS KIDS CHEWABLE TABLETS 2 PACK SACHET
BioGaia® Protectis® Chewable Tablets for Kids is a probiotic to help support immunity and for digestive health in growing children.
...Allergies
First Aid
-
$0.00
CRYSTADERM
Crystaderm® First Aid Cream provides strong and effective protection against minor skin infections, with no known risk of bacterial resistance1.
...Diabetes
-
$0.00
CareSens Dual Blood Glucose Meter
The CareSens Dual Blood Glucose/Ketone Meter is a compact, easy-to-use device that measures both blood glucose and ketone levels from a single meter, ideal for diabetes and ketone management.
ALWAYS READ THE LABEL AND...
$0.00
CareSens N Blood Glucose Meter
The CareSens N blood glucose meter is a great choice for people who would prefer a bigger screen and larger numbers. Its cleverly curved body design also helps make it easy to hold.
$0.00
CareSens N Pop Blood Glucose Meter
The CareSens N POP blood glucose meter is perfect for those who need to test frequently or who would prefer a small, slim meter that is easy to slip into a purse or pocket.
...$0.00
CareSens N Premier Blood Glucose Meter
The CareSens N Premier blood glucose meter helps make managing your data easy. With Bluetooth functionality, it provides quick and easy transfer of your readings to the SmartLog app on your compatible phone.
ALWAYS...
$0.00
CareSens N Voice Blood Glucose Meter
The meter that talks! With its talking function, the CareSens N Voice makes testing a little bit easier. It also has a large screen and numbers making it a great choice for those with...
00Your CartYour cart is empty -
-
-
-
-
-
-
-
-